Enclosing ketamine
How patents built the Spravato monopoly
In a previous post, I examined the political economy of ketamine as a mental health treatment. There I showed how patients in the United States are squeezed between two bad options: an expensive and less effective proprietary formulation in Spravato, and generic clinics that stay afloat by cutting care.
I argued that this outcome reflects what Thorstein Veblen called strategic sabotage. In this view, profitability depends not on wellbeing or therapeutic efficacy, but on restricting access and narrowing the scope of care. Even across very different market structures—monopoly in drug development and fragmented competition in clinical delivery—the same logic prevails.
Strategic sabotage is most visible in drug development. Pharmaceutical “inventions” rest on a communal stock of scientific and clinical knowledge built up over generations of shared research and experimentation. Patents are, in Lewis Mumford’s words, “a convenient falsehood” that allow companies to enclose this collective inheritance by claiming exclusive rights over the final incremental step in a much larger process of knowledge production.
As we saw, Spravato illustrates the pattern clearly. Johnson & Johnson (J&J) isolated one of ketamine’s isomers, packaged it as a nasal spray, and secured patents and regulatory exclusivity. Those legal restrictions transformed a cheap generic drug into a privately controlled asset, one that became psychiatry’s first blockbuster in decades.
In this post, I take a closer look at the ketamine patent landscape. I show how control over knowledge is concentrated, and how that concentration mirrors the financial dominance of J&J discussed in the previous post.
To analyse the ketamine patent landscape, I first identified patents related to ketamine and its enantiomers that were filed by for-profit companies developing ketamine-based treatments for mental health. To do this, I used Psychedelic Alpha’s Ketamine Patent Tracker to identify firms with active ketamine drug development programmes.
This search yielded 67 patents filed by 18 commercial entities, including 16 drug developers and two joint ventures between drug developers. I also added the landmark ketamine patent held by Yale University and licensed to Janssen, the wholly owned subsidiary of J&J that developed Spravato.
Of the 67 patents in the dataset, J&J (including the Yale-licensed patent) accounts for 25, or 37% of the total. Looking only at granted patents, J&J holds 10 of the 29, a 34% share. Even on this simple patent count, Johnson & Johnson already stands out as the dominant player.
But a simple patent count can only take us so far. It tells us little about the influence of those patents. One way to get at this is to look at the citations those patents receive. As part of a patent application, the applicant must disclose “prior art” to the patent office. Prior art is any existing knowledge or invention that is relevant to the new claim. Applicants disclose it by citing earlier patents and relevant non-patent literature, with patent examiners often supplementing these citations during the review process.
In both mainstream and critical studies of innovation, patent citations are often treated as a proxy for innovation. Put simply, backward citations (who you cite) show how incremental an invention is, while forward citations (who cites you) show novelty. In a power-centred approach, patent citations instead measure control over the technological landscape. They are indicators of sabotage. To cite a patent is to recognise a competitor’s legally sanctioned power of exclusion.
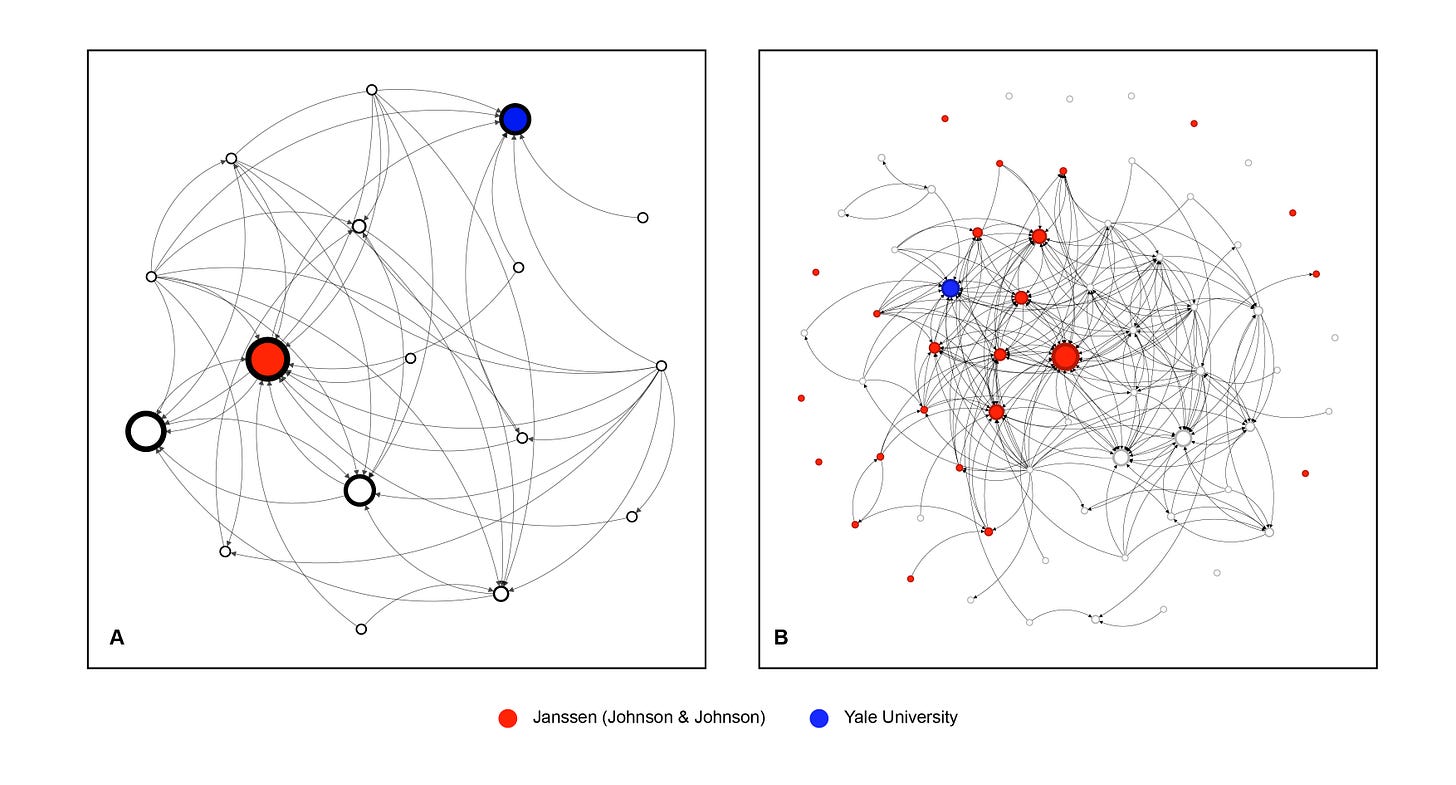
Figure 1 shows the ketamine patent citation network at both the company level (panel a) and the patent level (panel b). Node size reflects PageRank, a measure of influence that captures not only how often a firm or patent is cited, but also whether those citations come from other influential nodes. Janssen’s patents are shown in red, while the blue node represents the Yale esketamine patent licensed to Janssen.
Figure 1 The ketamine patent citation network
Note: Panel A shows the company-level network; Panel B shows the patent-level network. Node size reflects PageRank; edges represent citations. Network layouts are generated using a force-directed Fruchterman–Reingold algorithm to emphasize relational structure. Self-citations are excluded at the company level and included at the patent level.
Source: Psychedelia Alpha Ketamine Patent Tracker; Google Patents; author’s calculations and visualization using Gephi.
In panel A, Janssen stands out as the dominant hub, with citations flowing in from across the network. Most other firms appear as smaller, more weakly connected nodes arranged around this centre, forming a clear periphery. Yale also appears as a large and well-connected node, closely linked to the central cluster around Janssen.
In panel B, this structure becomes more pronounced. A dense core is organised around a handful of highly influential patents, almost all of them held by Janssen. The Yale patent is also within this core, heavily cited and closely connected to the same cluster. Beyond the core, the bulk of patents are small, weakly connected, and located at the periphery of the network.
The previous post showed J&J’s financial dominance in ketamine drug development. Here, we see its knowledge dominance. The two go hand in hand. Control over profits rests on control over knowledge.
There is no point moralising over what J&J has done with ketamine as a mental health treatment. The company is just playing by the rules of drug development: securing monopoly control through patents and turning it into blockbuster returns.
And there’s no reason to expect psychedelic companies to act differently. As drug developers, they operate under the same constraints. For all the talk about disrupting Big Pharma, they end up following the same playbook, building “IP moats” and using Spravato as their blueprint for commercialisation.
If and when other psychedelics are approved, we should expect the same winner-take-most dynamics to surface. That likely means high-priced, patented “innovations” that provide little benefit over cheaper generic options. Again, the aim is not better treatments, but profitability secured through the control and restriction of access.